- GST No. 27AANFV2416Q1ZH
- Send SMS
- Send Email
| Business Type | Manufacturer, Supplier, Retailer, Wholesaler |
| CAS No. | 100-97-0 |
| Purity | 99.8% |
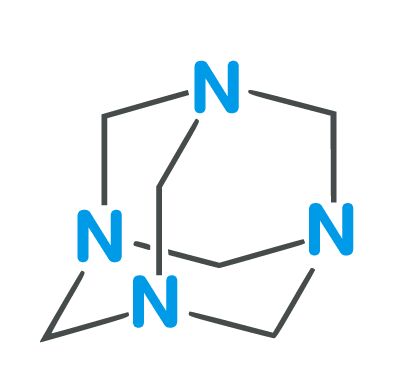
| Full Name | Hexamethylenetetramine |
| Click to view more | |
Preferred Buyer From
| Location | Anywhere in India |
Product Details
Formula
C6H12N4
Molar mass
140.186 g/mol
Melting point
280 °C (536 °F; 553 K)
Water
≤0.5%
Sulphate (SO4)
≤0.02%
Uses :
It is used in the preparations of phenolic (phenolic formaldehyde) resins and phenolic resin molding compound, where it is added as a hardening component. These products are used as binders, e.g., in brake and clutch linings, abrasive products, no-woven textiles, fireproof materials. Phenolic formaldehyde resins include synthetic thermosetting resins got by the reaction of phenol (an organic compound) and formaldehyde.
- Medicinal uses :
Medical professionals use hexamine as an antibiotic to treat urinary tract infections. The specific medication used is called methenamine hippurate, which is formed by the combination of hexamine and hippuric acid. In acidic urine, methenamine hydrolyzes into ammonia and formaldehyde, a non-specific antibiotic. As this reaction is activated by acid in the urine, methenamine is often ineffective as an antibiotic in those whose urine is not acidic. - Combustion :
Hexamine is also the primary ingredient in Esbit® fuel tablets. These fuel tablets have a very high energy density, burn smokeless and leave no ashes which also do not liquefy. They are used for cooking purposes by campers and military organizations. Esbit® fuel tablets are very lightweight and portable, but the heat they produce is difficult to control and the fumes it produces are noxious. Some people are allergic to the compound, which can be absorbed through the skin. - In fire-protection laboratories :
Hexamine tablets are used by fire-protection laboratories as a clean and reproducible fire source to test the flammability of carpets and rugs. - As a chemical agent in organic chemistry :
Organic chemists use hexamine as a chemical agent in several chemical reactions which are quite complex and is often used for teaching purposes. Very often they are used for purposes of chemical analysis to determine the identity of an unknown solute (particles that are dissolved into a solution) in a solution. - Other uses :
Hexamine is also used in rubber and textile adhesives, in paints and lacquers, in the photographic industry, in the production of deodorants and hair fixers.